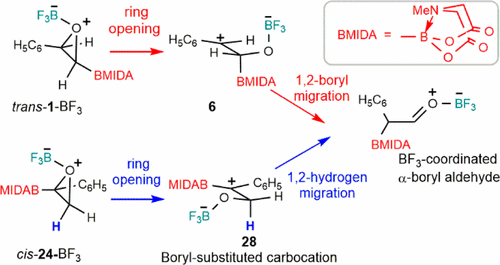
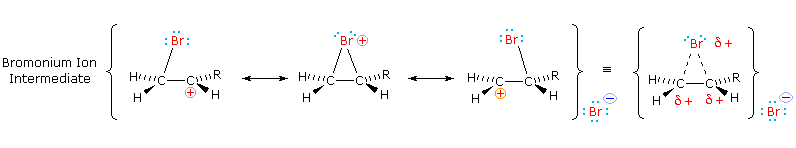
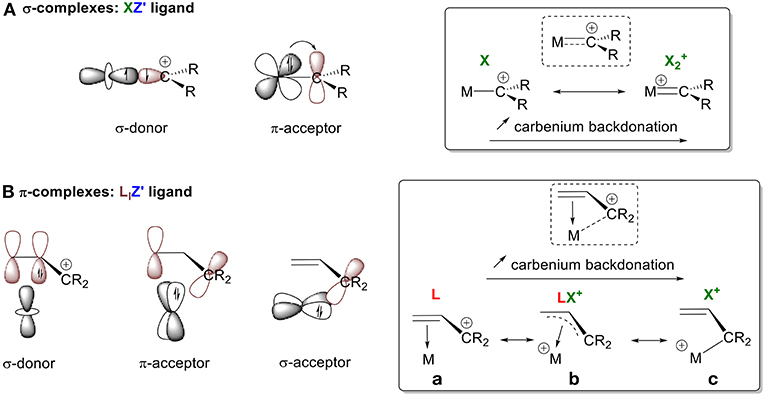
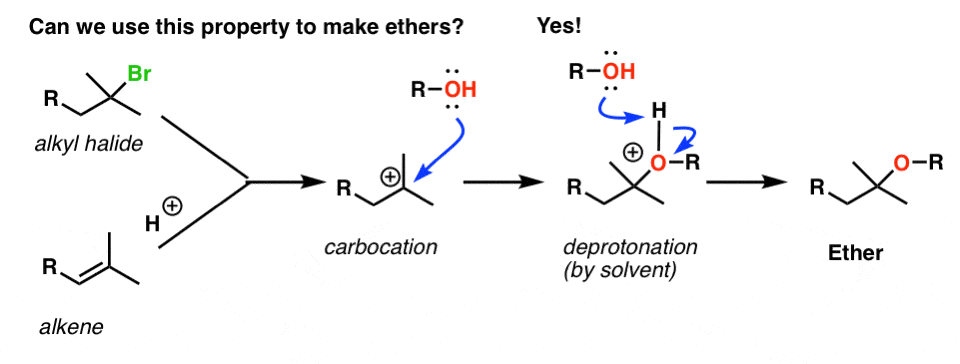
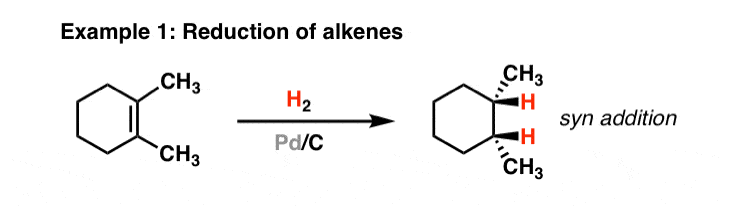
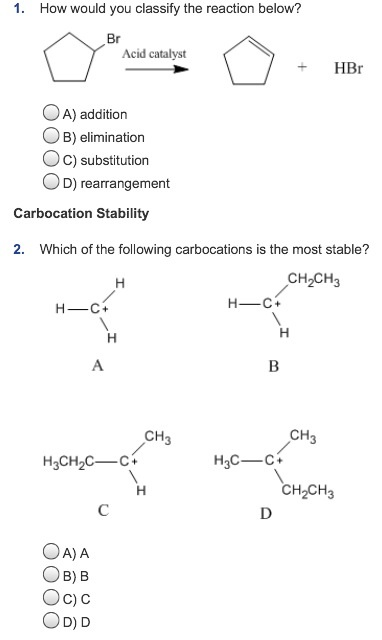
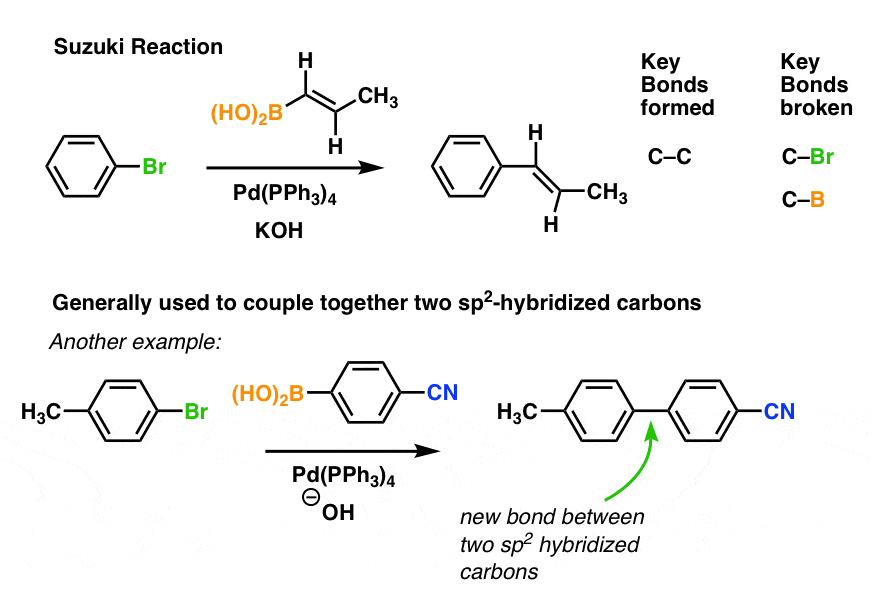
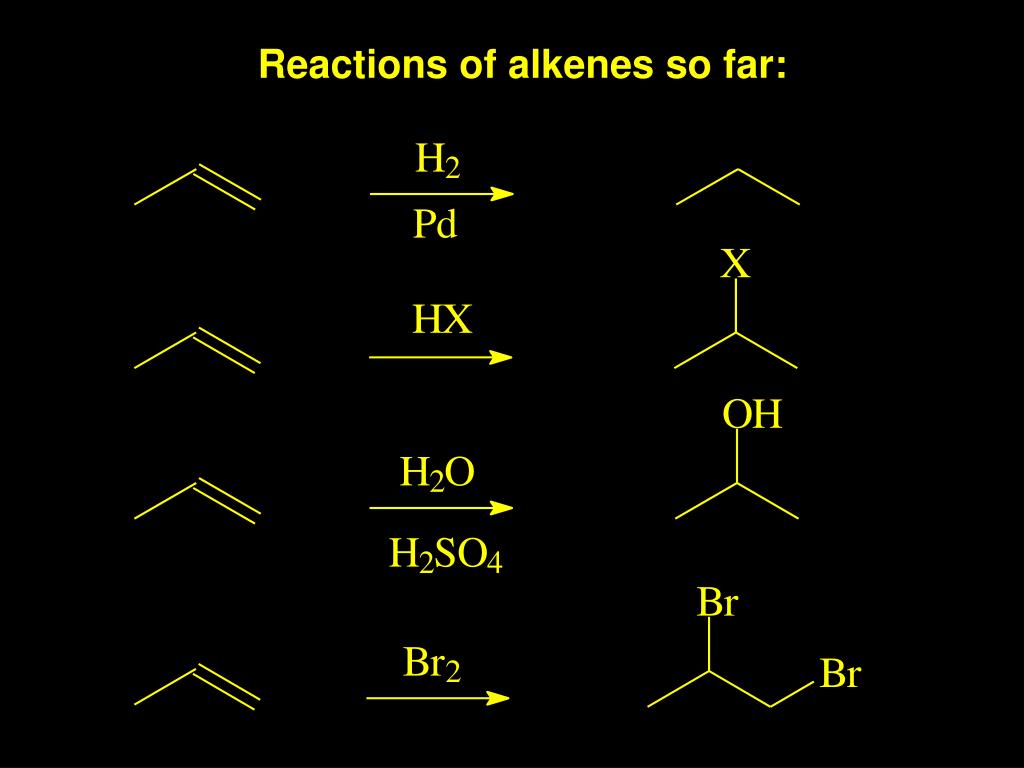
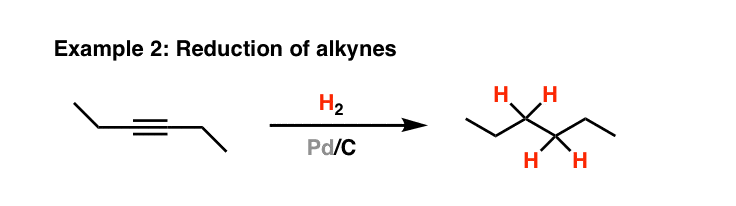
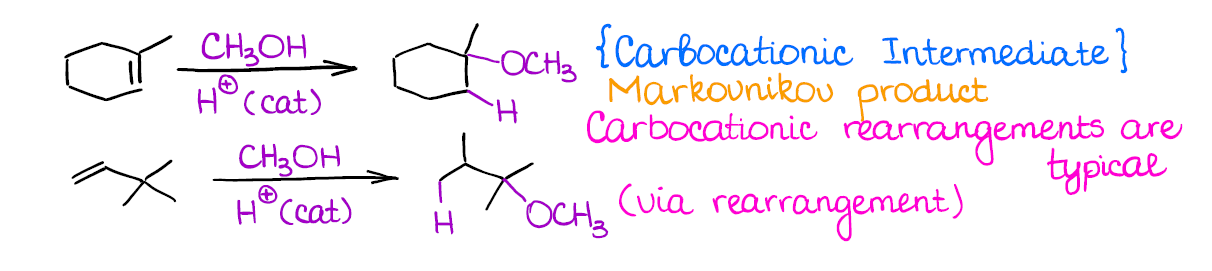
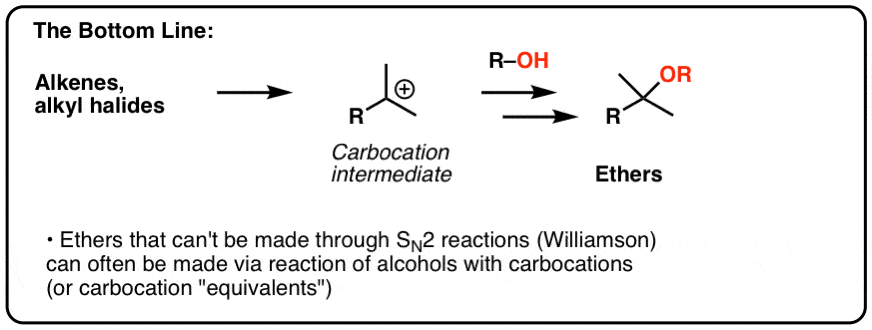
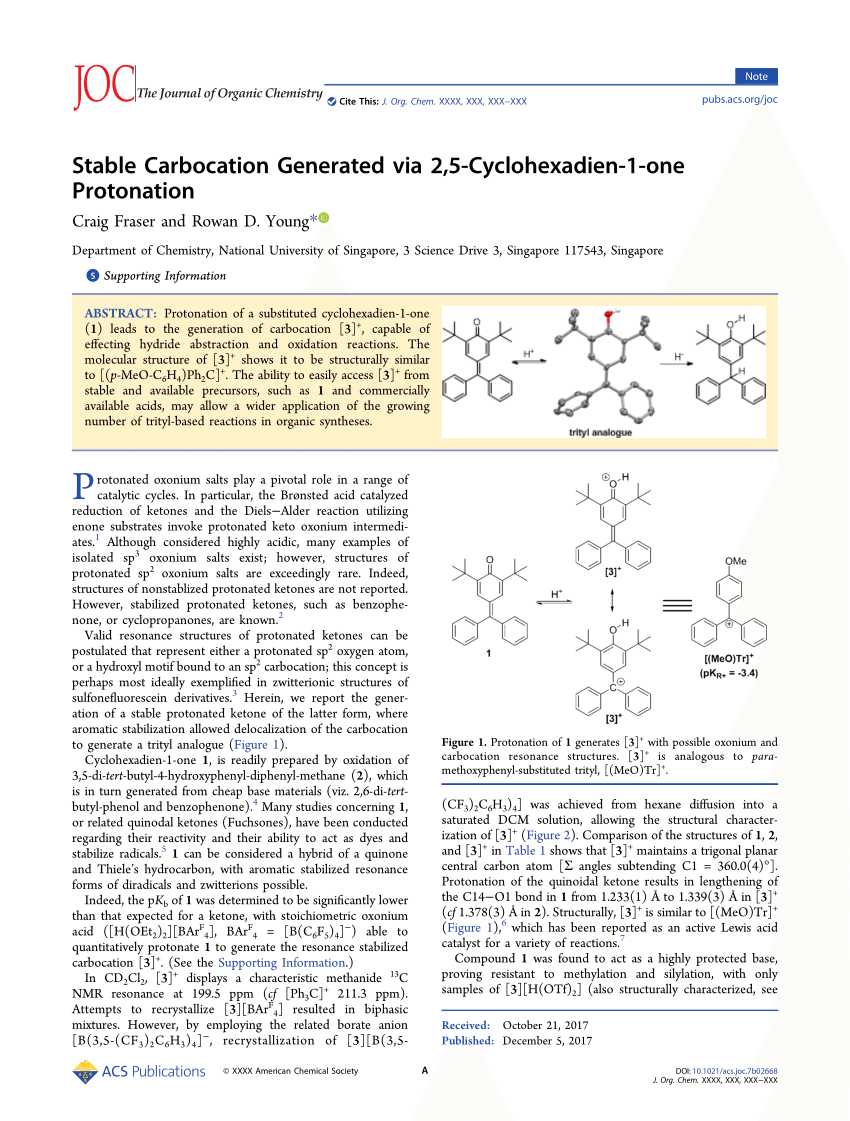
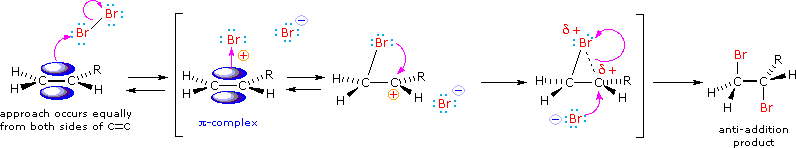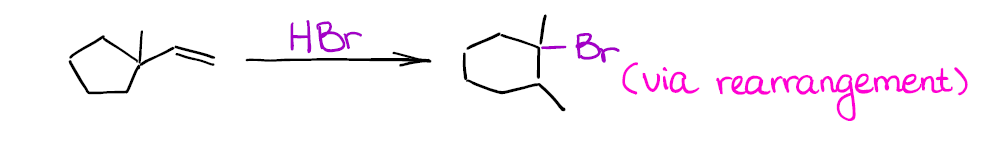Tailored Cationic Palladium( 11) Compounds as Catalysts for Highly Selective Dimerization and Polymerization of Vinylic Monomers
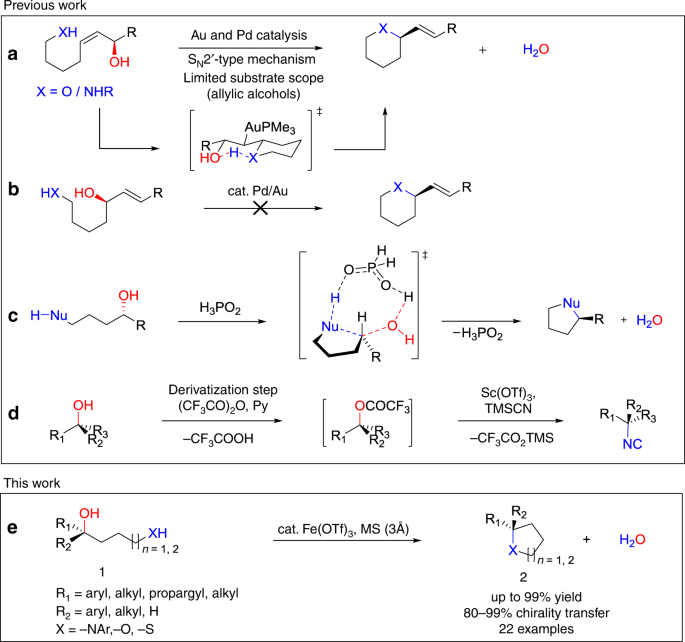
Intramolecular substitutions of secondary and tertiary alcohols with chirality transfer by an iron(III) catalyst | Nature Communications
![Table 2 from The air-stable carbocation salt [(MeOC6H4)CPh2][BF4] in Lewis acid catalyzed hydrothiolation of alkenes. | Semantic Scholar Table 2 from The air-stable carbocation salt [(MeOC6H4)CPh2][BF4] in Lewis acid catalyzed hydrothiolation of alkenes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a6907c824f2033c2744d5d7f554a45f911d8dffc/2-Table2-1.png)
Table 2 from The air-stable carbocation salt [(MeOC6H4)CPh2][BF4] in Lewis acid catalyzed hydrothiolation of alkenes. | Semantic Scholar

Chiral Anion Directed Asymmetric Carbocation‐Catalyzed Diels–Alder Reactions - Eur. J. Org. Chem. - X-MOL
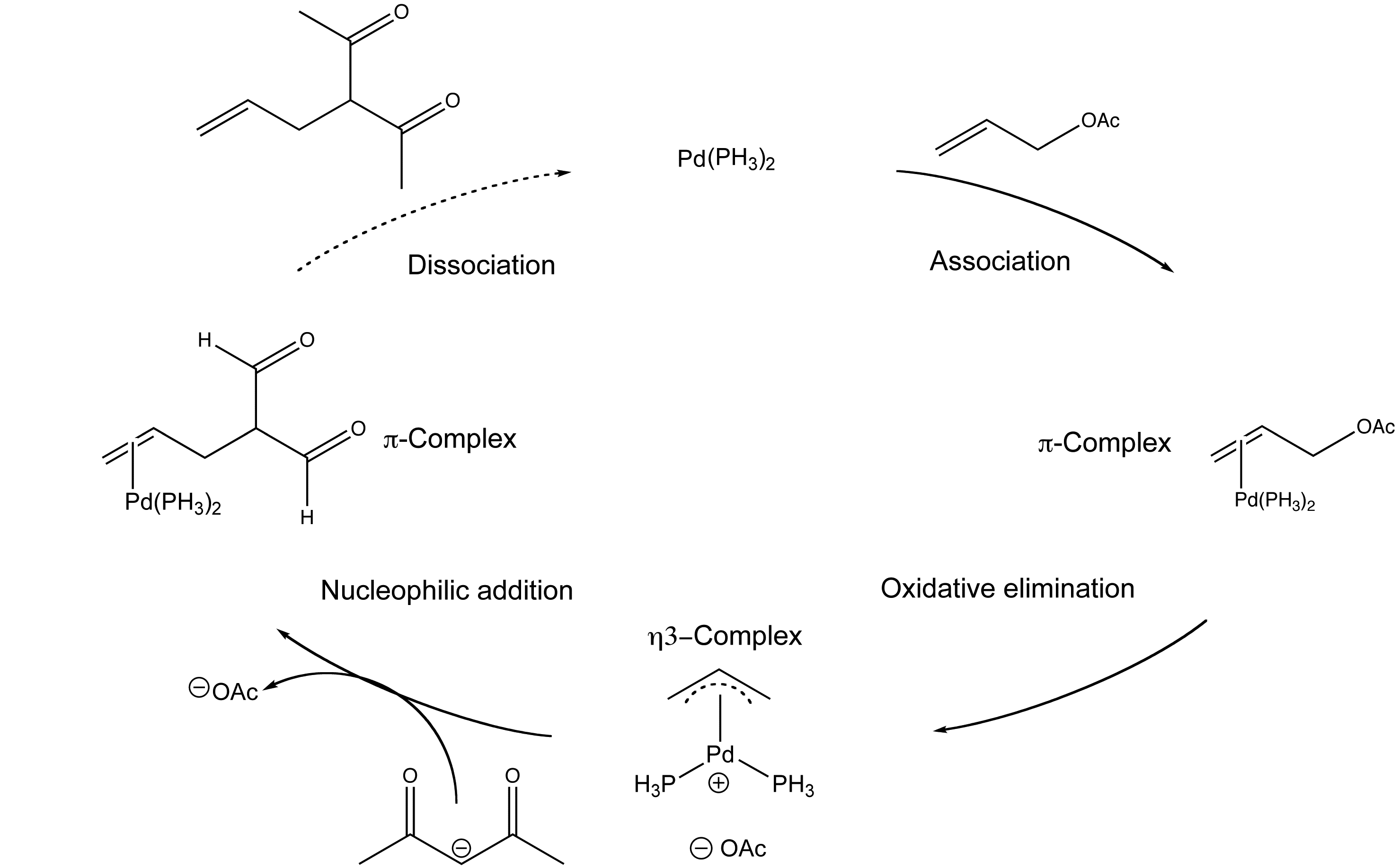
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

In(OTf)3‐Catalyzed Highly Chemo‐ and Regioselective Head‐to‐Tail Heterodimerization of Vinylarenes with 1,1‐Diarylethenes - Dai - 2011 - Chemistry – A European Journal - Wiley Online Library

Triazine-Based Cationic Leaving Group: Synergistic Driving Forces for Rapid Formation of Carbocation Species,The Journal of Organic Chemistry - X-MOL

Stereoselective Reactions with Stabilized Carbocations - Cozzi - 2010 - Angewandte Chemie International Edition - Wiley Online Library

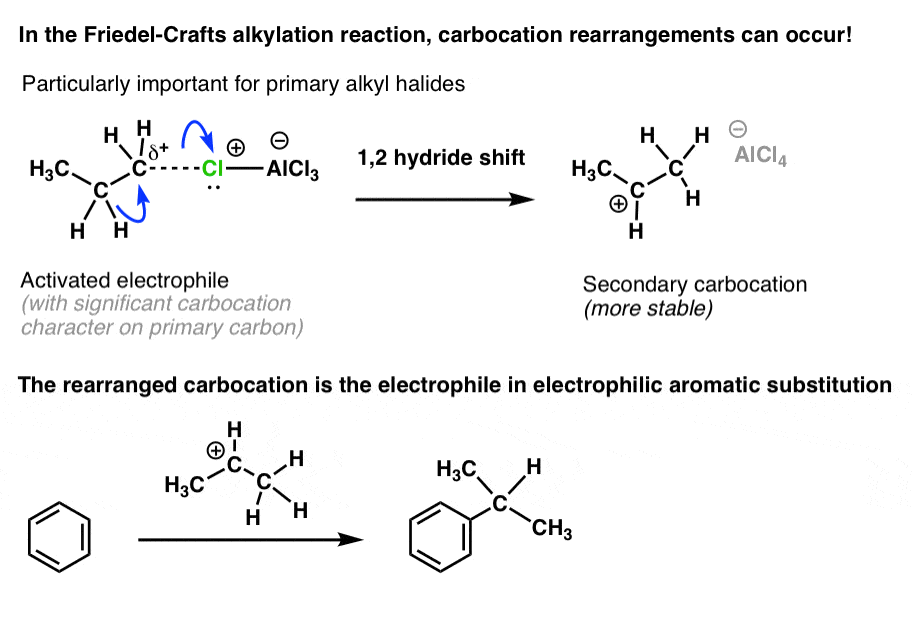
Insight into the shift and rearrangement of carbocation in Friedel-Crafts alkylation of unsaturated fatty acids revealed by GC–MS - ScienceDirect