Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML
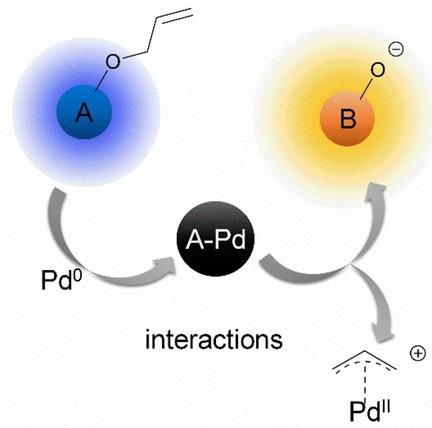
Kinetics of Palladium(0)‐Allyl Interactions in the Tsuji‐Trost Reaction, derived from Single‐Molecule Fluorescence Microscopy - ChemCatChem - X-MOL
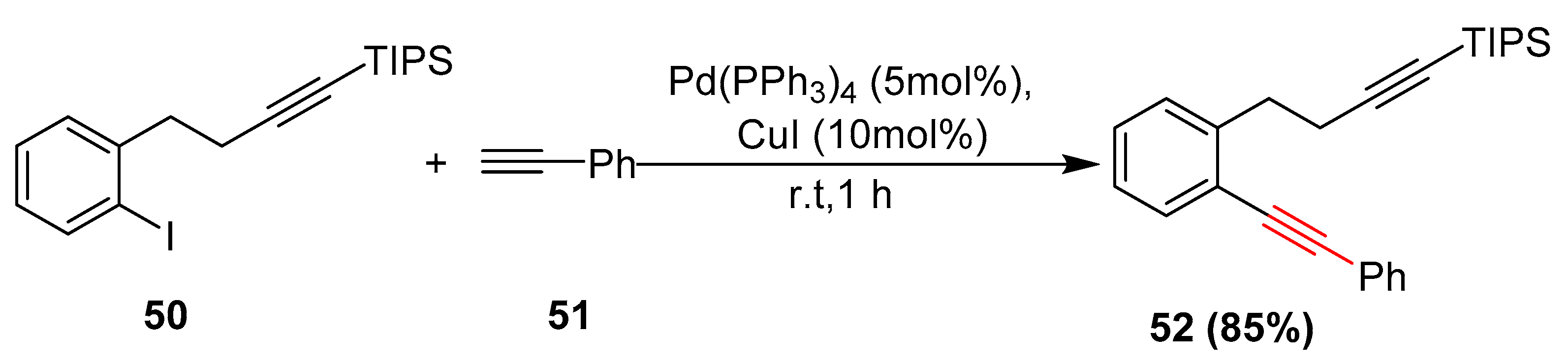
Catalysts | Free Full-Text | Palladium and Copper Catalyzed Sonogashira cross Coupling an Excellent Methodology for C-C Bond Formation over 17 Years: A Review | HTML

Recent Advances in Pd‐Catalyzed Cross‐Coupling Reaction in Ionic Liquids - Li - 2018 - European Journal of Organic Chemistry - Wiley Online Library
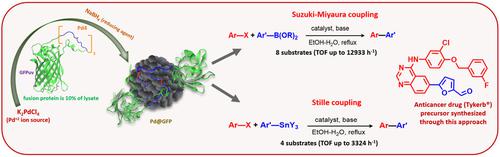
Recombinant Peptide Fusion Protein‐Templated Palladium Nanoparticles for Suzuki‐Miyaura and Stille Coupling Reactions - ChemCatChem - X-MOL
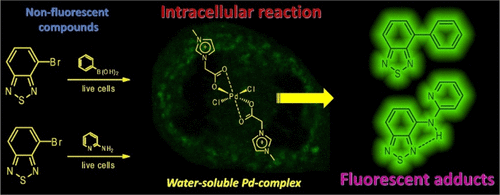
Palladium Catalyst with Task-Specific Ionic Liquid Ligands: Intracellular Reactions and Mitochondrial Imaging with Benzothiadiazole Derivatives. - J. Org. Chem. - X-MOL
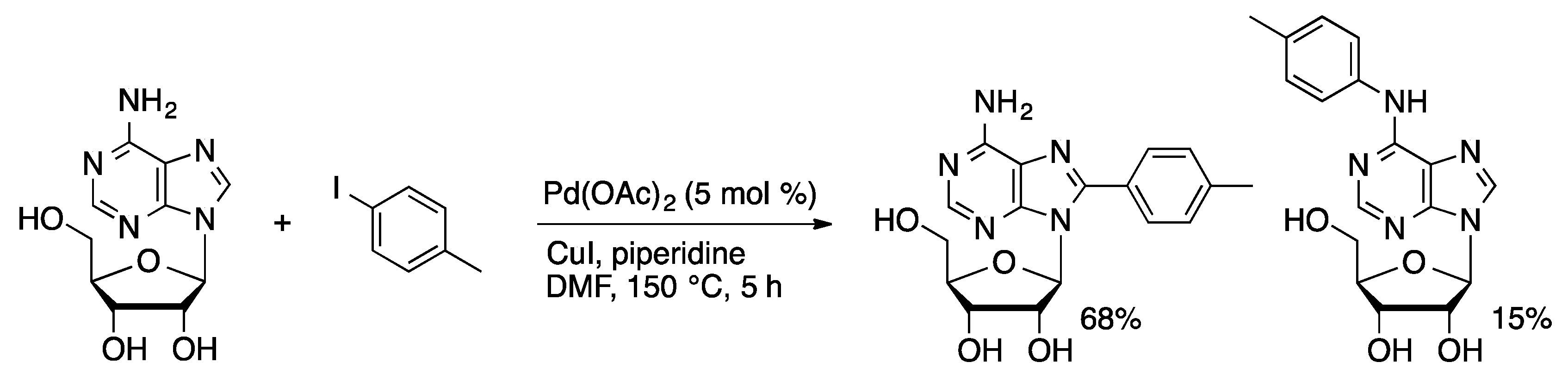
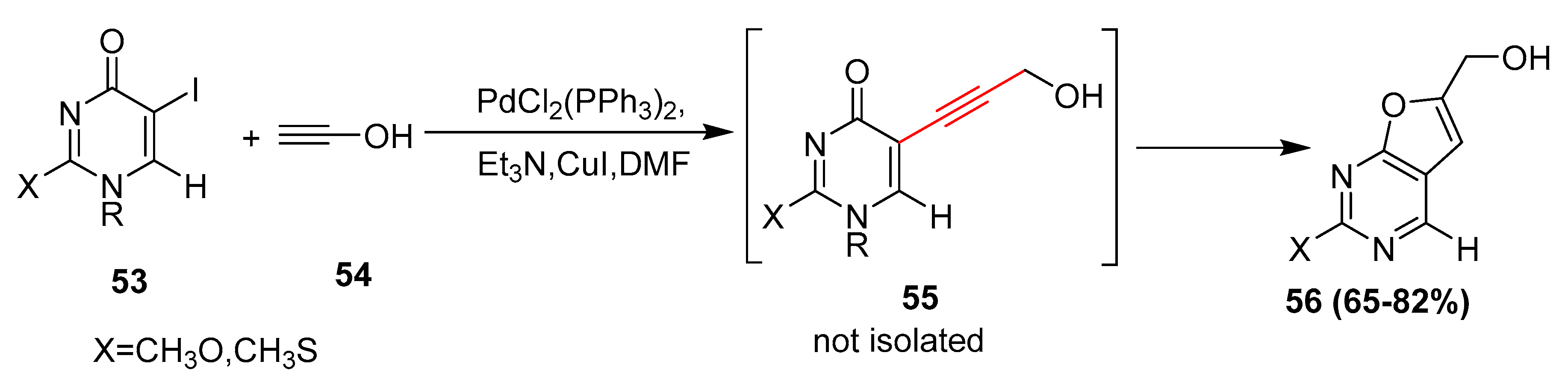
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

A highly selective fluorescent probe for the detection of palladium(II) ion in cells and aqueous media | SpringerLink

A novel colorimetric and fluorescent probe for the highly selective and sensitive detection of palladium based on Pd(0) mediated reaction - ScienceDirect

Synthetic route of molecules 6, 7 and 8 through a palladium-catalyzed... | Download Scientific Diagram

Atomically dispersed palladium catalyses Suzuki–Miyaura reactions under phosphine-free conditions | Communications Chemistry
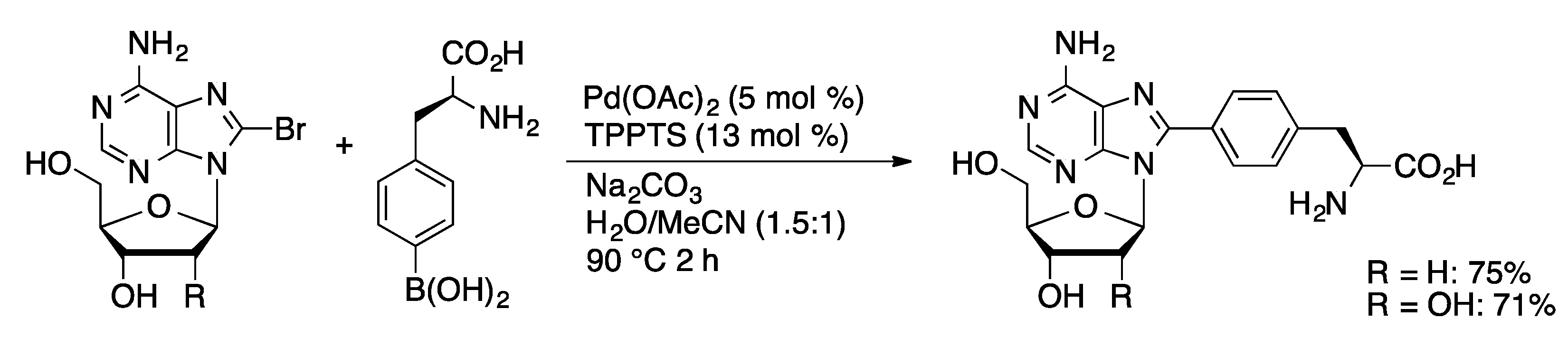
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

An umbelliferone-derivated fluorescent sensor for selective detection of palladium(II) from palladium(0) in living cells - ScienceDirect
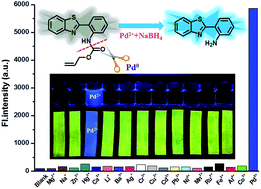
A highly sensitive and selective fluorescent probe based on a Pd-catalyzed reaction for detection of Pd2+† - Anal. Methods - X-MOL

Palladium‐Catalyzed Coupling Reactions for the Functionalization of BODIPY Dyes with Fluorescence Spanning the Visible Spectrum - Rohand - 2006 - European Journal of Organic Chemistry - Wiley Online Library
![PDF] A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water | Semantic Scholar PDF] A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ba172da9f8cadb8e5a159ea6f4d3f841314544f5/6-Figure2-1.png)
PDF] A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water | Semantic Scholar
A Fluorescence Active Catalyst Support Comprising Carbon Quantum Dots and Magnesium Oxide Doping for Stabilization of Palladium

Chemosensors | Free Full-Text | Sequential Detection of Palladium and Chromium Oxyanion by a Fluorescein Based Chemosensor in Mixed Aqueous Media | HTML
![The synthesis of fluorescent benzofuro[2,3- c ]pyridines via palladium- catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO01491H The synthesis of fluorescent benzofuro[2,3- c ]pyridines via palladium- catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO01491H](https://pubs.rsc.org/image/article/2020/QO/c9qo01491h/c9qo01491h-s1_hi-res.gif)
The synthesis of fluorescent benzofuro[2,3- c ]pyridines via palladium- catalyzed heteroaromatic C–H addition and sequential tandem cyclization - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO01491H

Figure 4 from A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water
1 Recent Advances in the Palladium Catalyzed Suzuki-Miyaura Cross-Coupling Reaction in Water Anamitra Chatterjee,[a] and Thomas
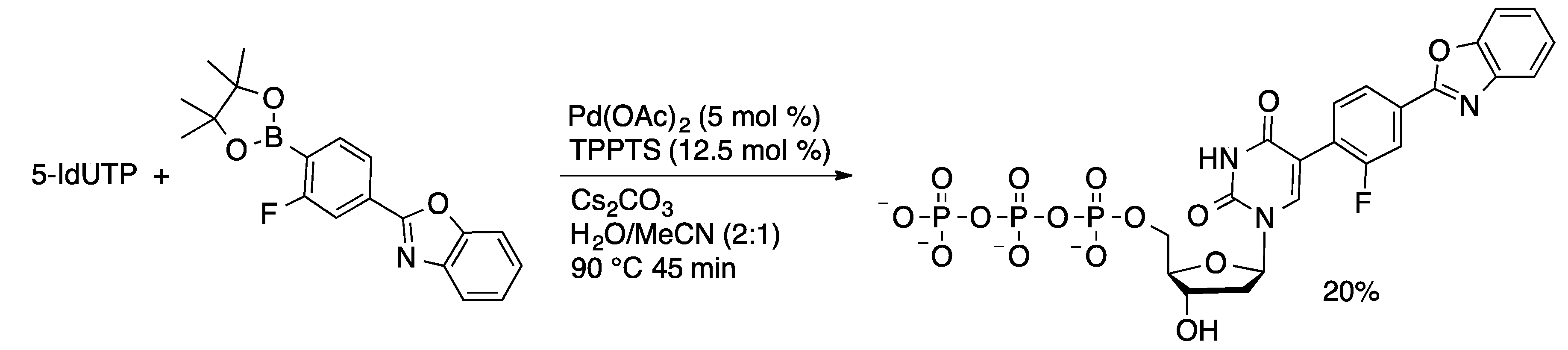
Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML

Molecules | Free Full-Text | The Current Status of Heterogeneous Palladium Catalysed Heck and Suzuki Cross-Coupling Reactions | HTML
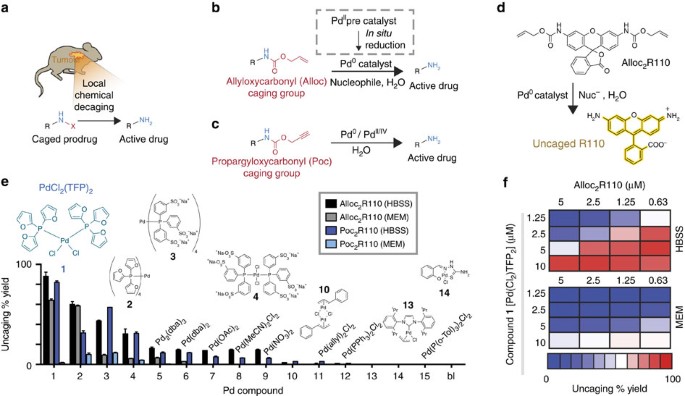
![Suzuki cross-coupling reactions catalysed by Pd-NPs in ILs [57]. a | Download Scientific Diagram Suzuki cross-coupling reactions catalysed by Pd-NPs in ILs [57]. a | Download Scientific Diagram](https://www.researchgate.net/profile/Jairton_Dupont/publication/45288394/figure/tbl1/AS:667217494556681@1536088455330/Suzuki-cross-coupling-reactions-catalysed-by-Pd-NPs-in-ILs-57-a.png)