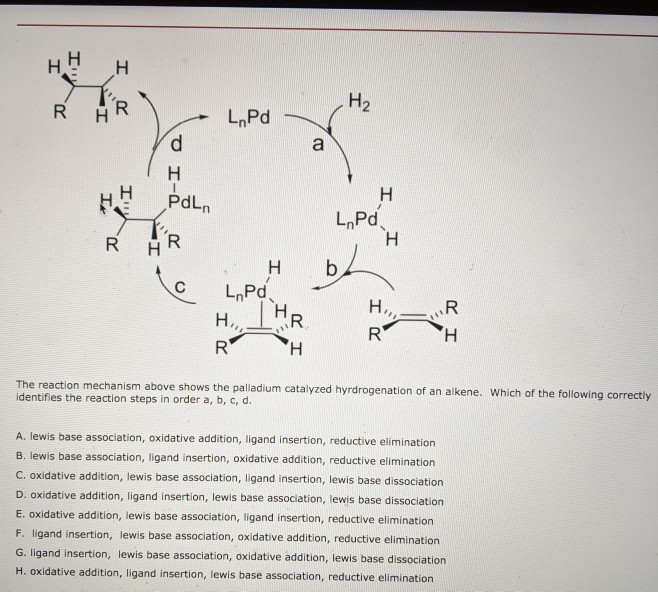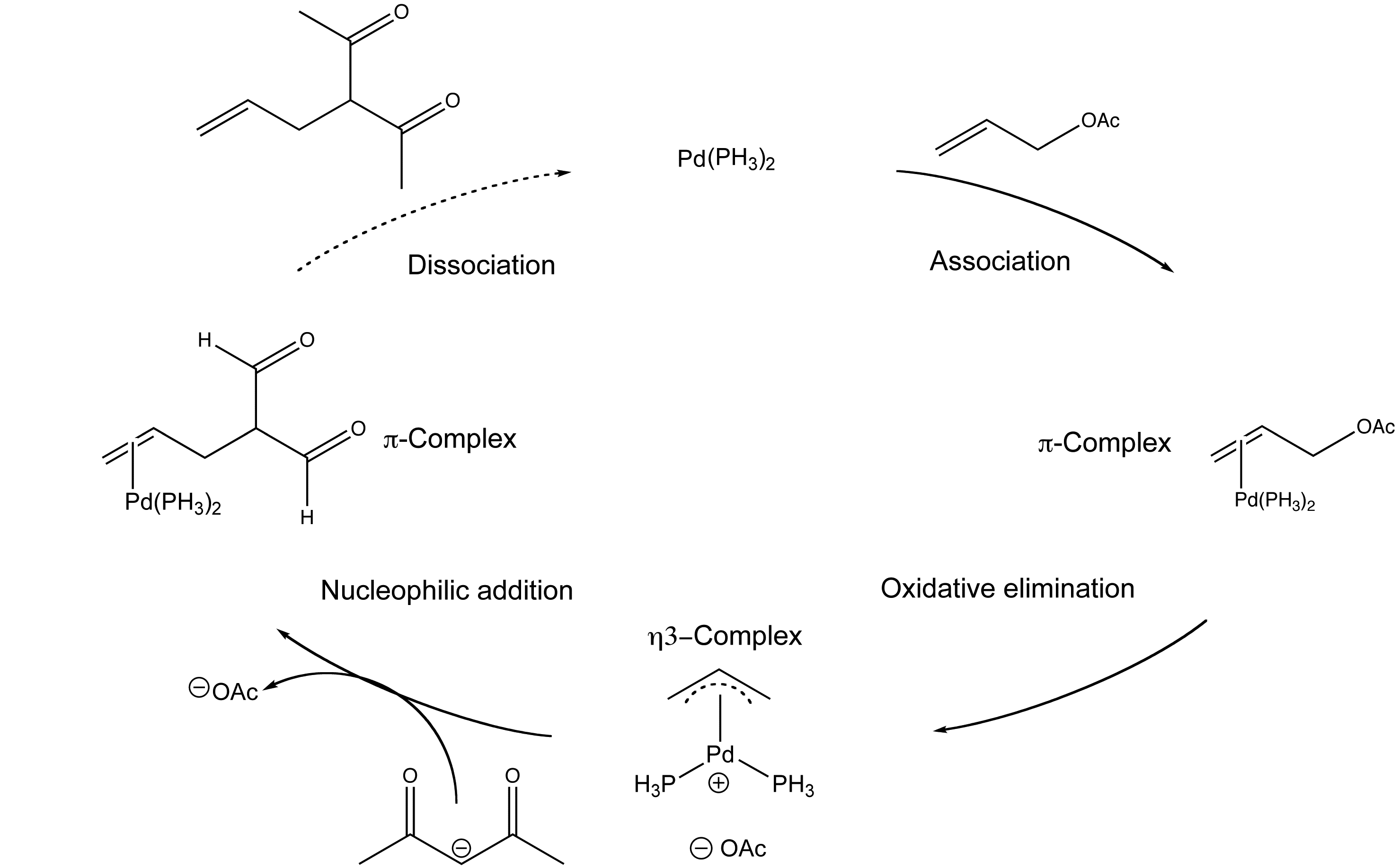
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds
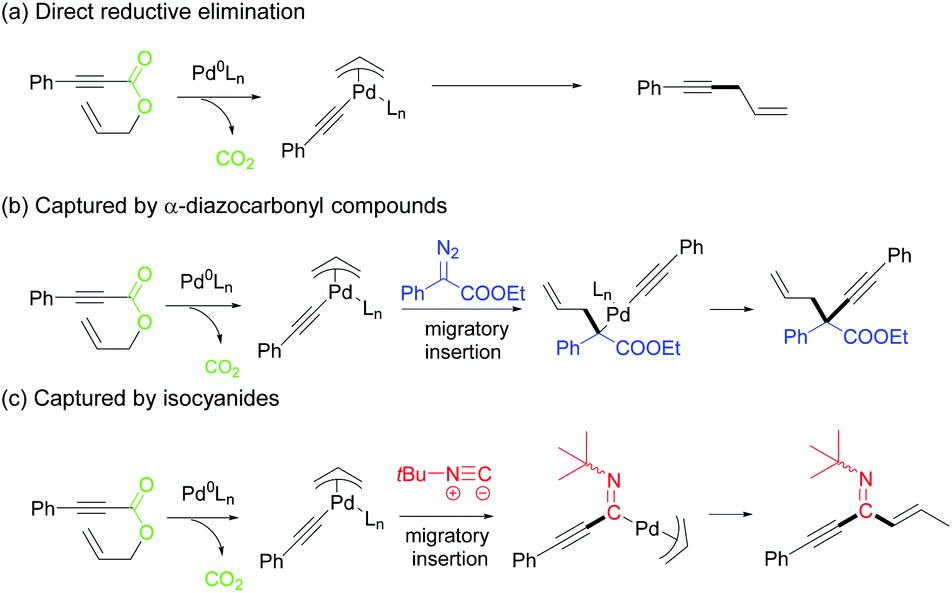
Synthesis of 1,4-enyne-3-ones via palladium-catalyzed sequential decarboxylation and carbonylation of allyl alkynoates - Organic Chemistry Frontiers (RSC Publishing)
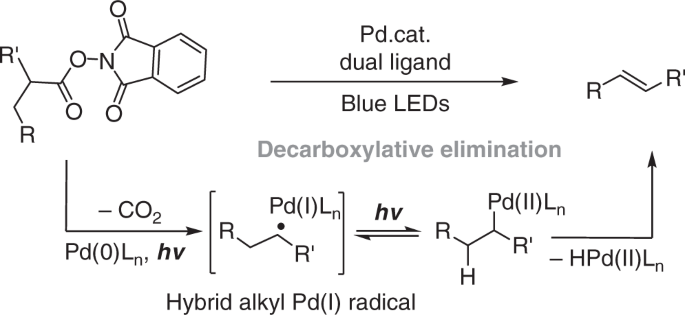
Irradiation-induced palladium-catalyzed decarboxylative desaturation enabled by a dual ligand system | Nature Communications

Mechanistic experiments support the proposed β-elimination mechanism... | Download Scientific Diagram
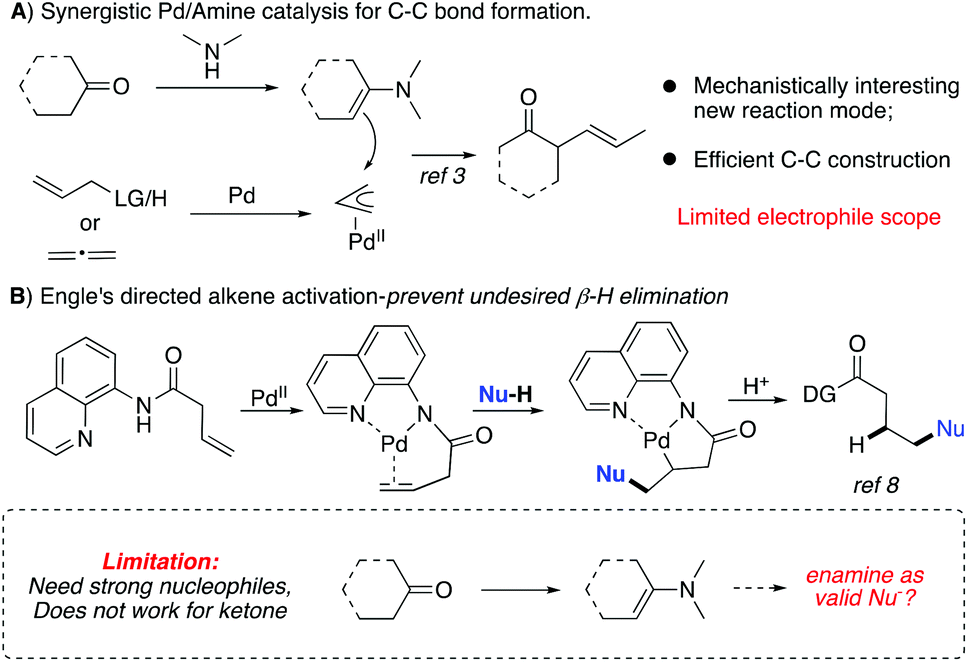
Synergistic palladium/enamine catalysis for asymmetric hydrocarbon functionalization of unactivated alkenes with ketones - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01165J
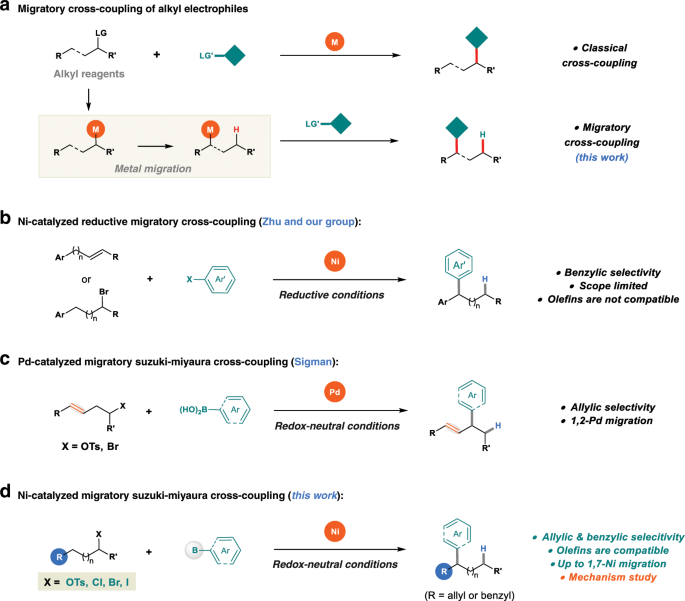
Reaction scope and mechanistic insights of nickel-catalyzed migratory Suzuki–Miyaura cross-coupling | Nature Communications

From Benzofurans to Indoles: Palladium‐Catalyzed Reductive Ring‐Opening and Closure via β‐Phenoxide Elimination - Perego - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

Activation of diverse carbon-heteroatom and carbon-carbon bonds via palladium(II)-catalysed β-X elimination | Request PDF

Palladium‐Catalyzed Elimination/Isomerization of Enol Triflates into 1,3‐Dienes - Crouch - 2011 - Angewandte Chemie International Edition - Wiley Online Library
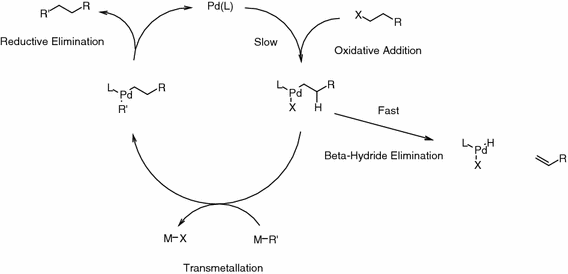
DFT studies of reductive elimination, C–H activation and β-hydride elimination in alkyl and aryl palladium amine complexes | SpringerLink

Figure 1 from Chemical remodeling of cell-surface sialic acids through a palladium-triggered bioorthogonal elimination reaction. | Semantic Scholar

Activation of diverse carbon–heteroatom and carbon–carbon bonds via palladium( ii )-catalysed β-X elimination | Nature Chemistry
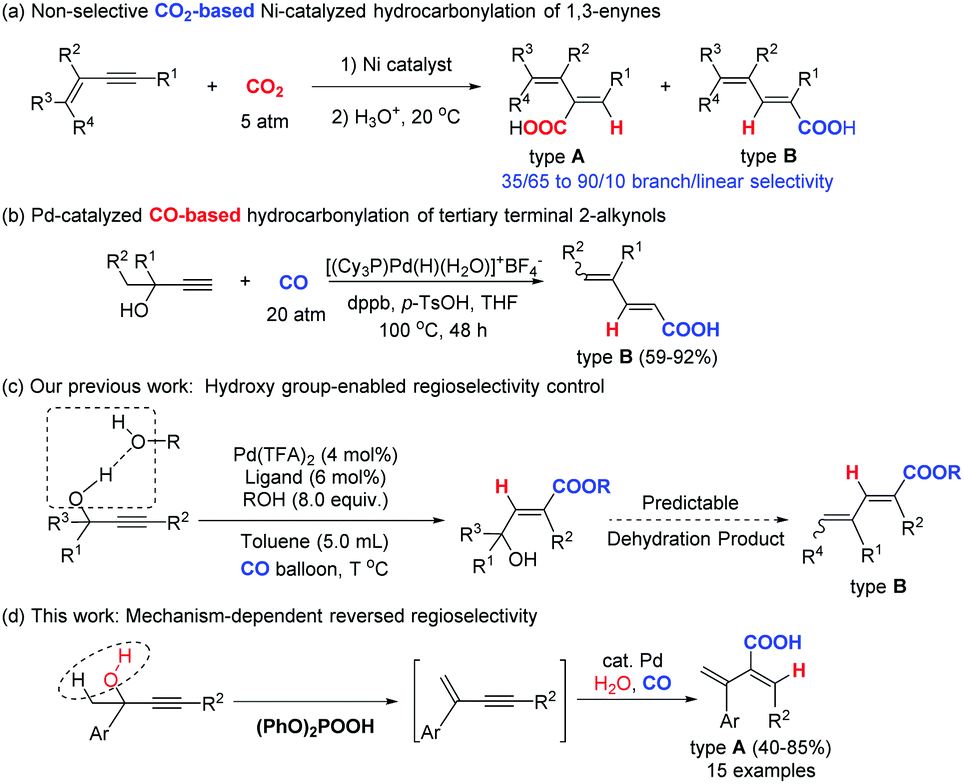
Palladium catalyzed regioselective elimination–hydrocarbonylation of propargylic alcohols - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC03262B