Molecules | Free Full-Text | The Current Status of Heterogeneous Palladium Catalysed Heck and Suzuki Cross-Coupling Reactions
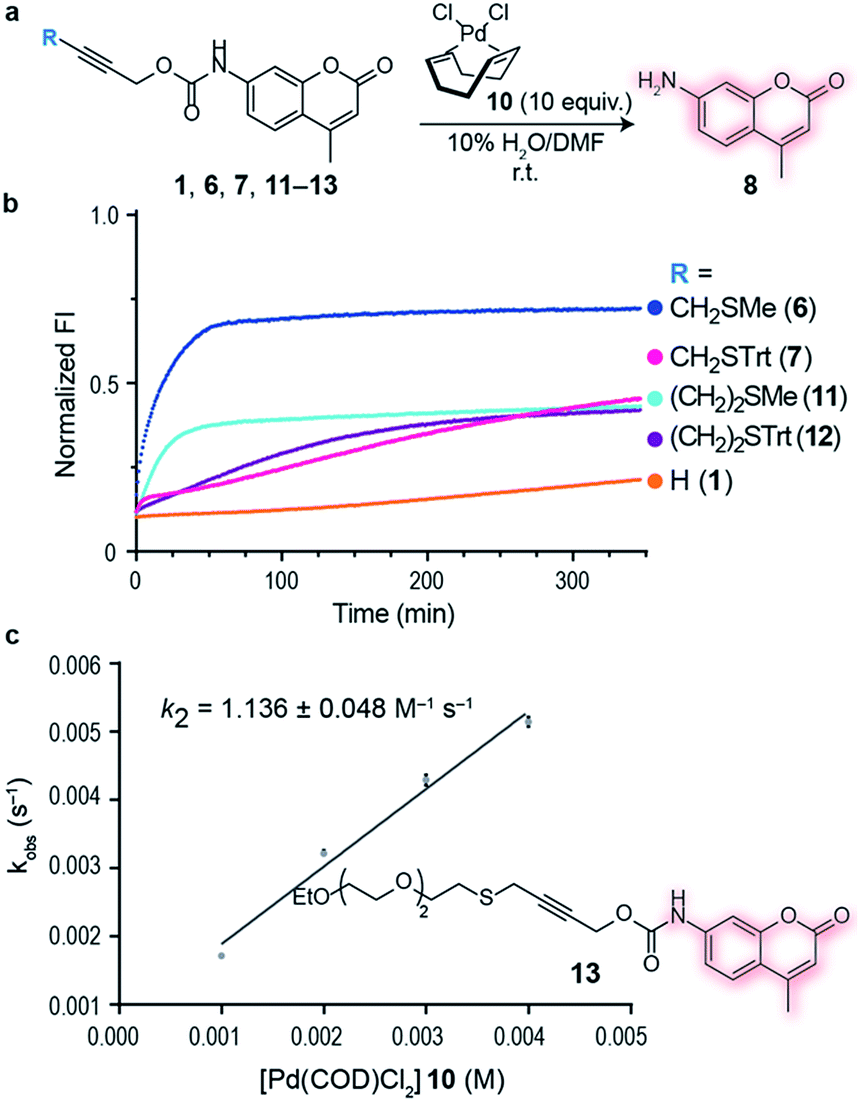
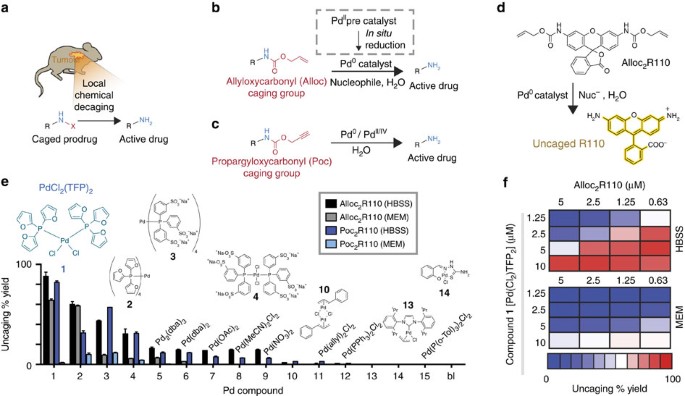
A thioether-directed palladium-cleavable linker for targeted bioorthogonal drug decaging - Chemical Science (RSC Publishing)

Close-to-Release": Spontaneous Bioorthogonal Uncaging Resulting from Ring-Closing Metathesis. - Abstract - Europe PMC
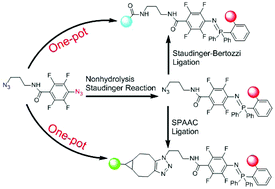
The one-pot nonhydrolysis Staudinger reaction and Staudinger or SPAAC ligation. - Org. Biomol. Chem. - X-MOL
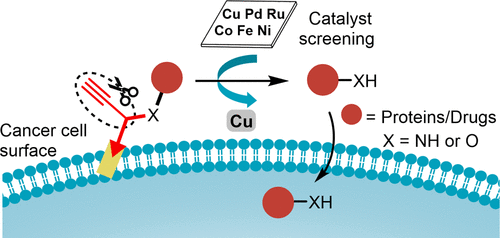
Copper-Triggered Bioorthogonal Cleavage Reactions for Reversible Protein and Cell Surface Modifications. - J. Am. Chem. Soc. - X-MOL
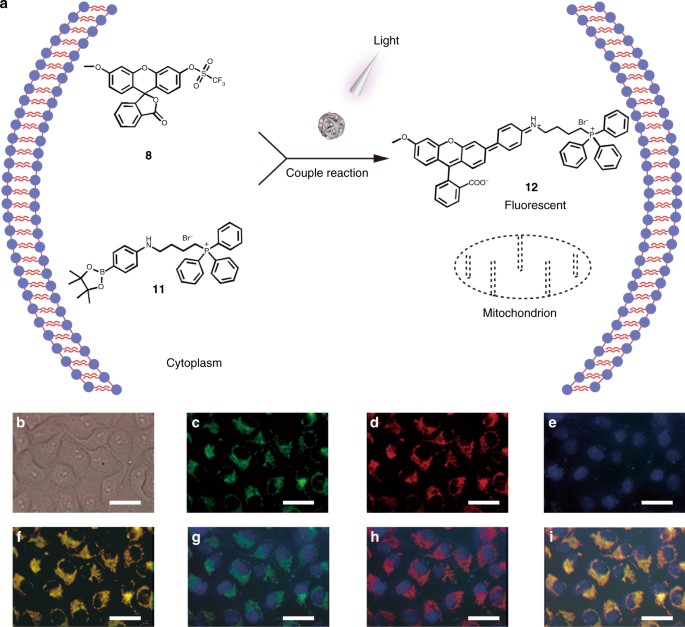
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications
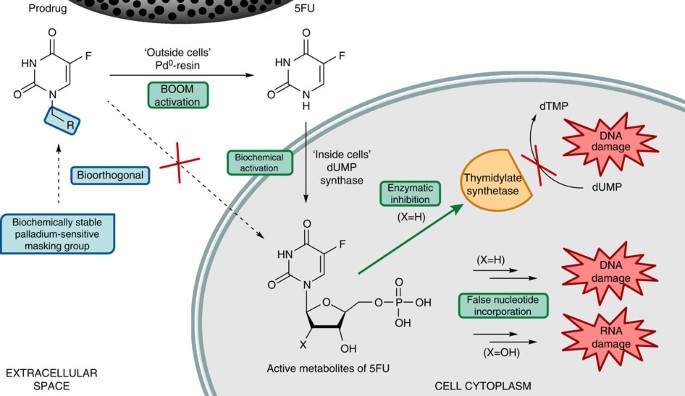
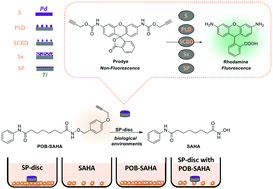
Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach | Nature Communications
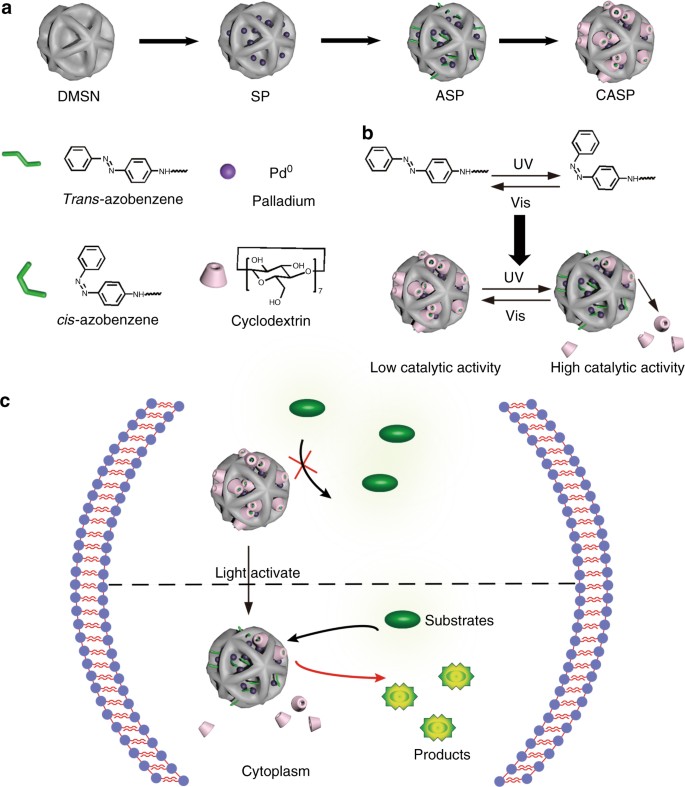
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications
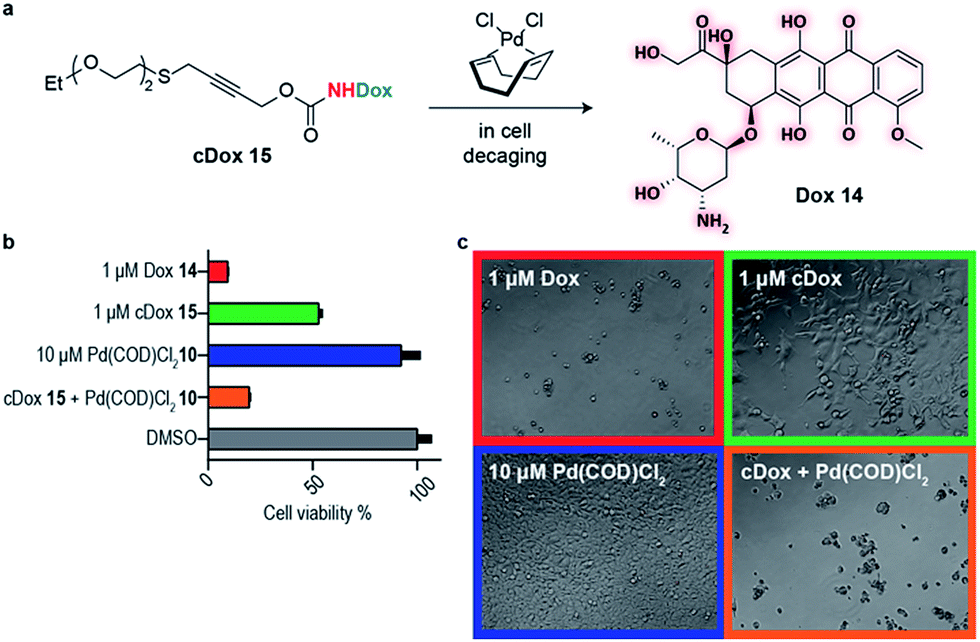
A thioether-directed palladium-cleavable linker for targeted bioorthogonal drug decaging - Chemical Science (RSC Publishing)

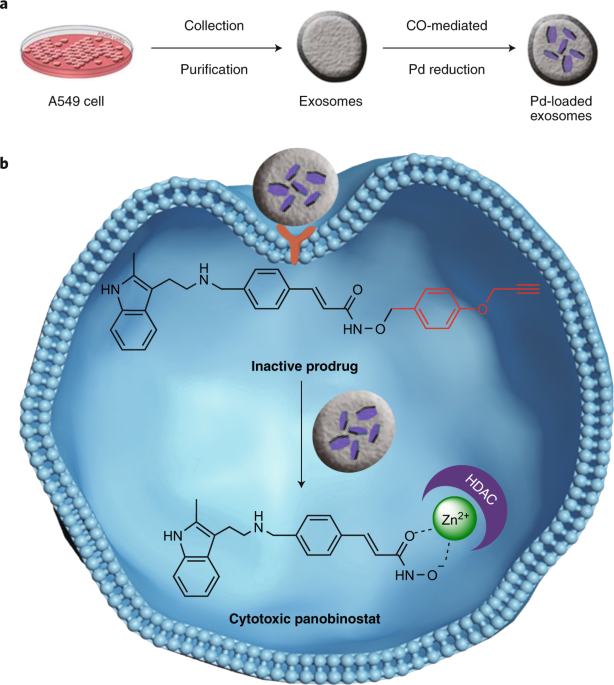
Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis | Request PDF
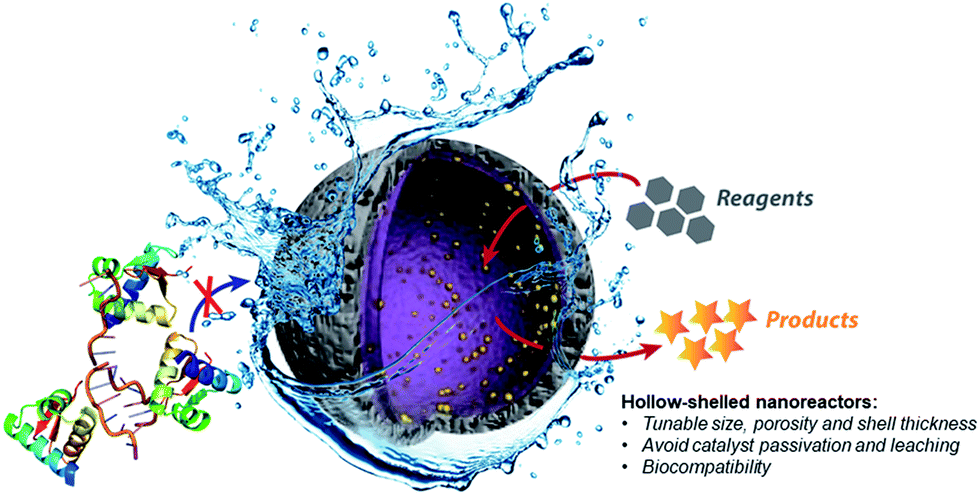
Hollow nanoreactors for Pd-catalyzed Suzuki–Miyaura coupling and O -propargyl cleavage reactions in bio-relevant aqueous media - Chemical Science (RSC Publishing) DOI:10.1039/C8SC04390F

Concurrent and orthogonal gold(I) and ruthenium(II) catalysis inside living cells | Nature Communications
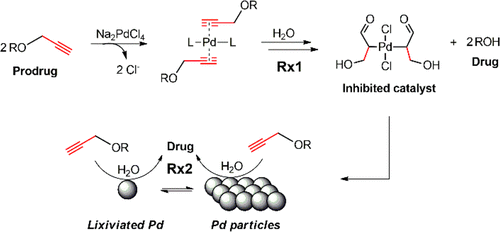
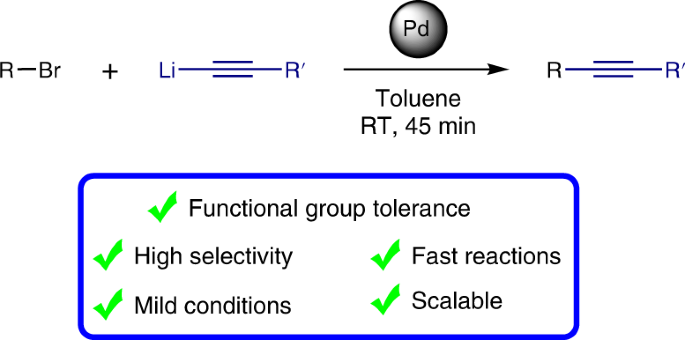
Mechanism of Palladium(II)-Mediated Uncaging Reactions of Propargylic Substrates,ACS Catalysis - X-MOL